Batteries are a means to transform chemical energy directly to electrical energy. A battery consists of a number of voltaic cells. Each cell is made up of two half-cells connected in series by a conductive electrolyte containing metal cations. One half-cell contains the electrolyte and the negative electrode, the electrode to which anions migrate, while the other half-cell includes the electrolyte and the positive electrode, to which cations migrate. In many cases, the electrical energy which is released is caused by the difference in the bond energies of the metals, oxides, or molecules when the electrochemical reaction takes place. Also, for many types of batteries, but particularly for lithium-ion batteries, the conditions under which they are produced have to be very dry in order for them to be able to hold large amounts of energy.
Now (2022), a team of scientists at Argonne National Laboratory has developed a new battery electrolyte which can contain a much greater amount of water than conventional electrolytes. The new battery electrolyte design was created using computer simulations of water in different electrolyte environments. The scientists found that the formulation they had created could contain a lot more water than previously suspected and could thus contribute to reducing costs in battery fabrication. By using an electrolyte made up of two kinds of salts, a lithium salt and an ionic liquid, the team created a medium in which much more water molecules could be stably absorbed by the electrolyte.
To support the experimental results and investigate the underlying chemical mechanisms, computer simulations were performed on the Theta supercomputer at Argonne analysing the electrolyte close to the electrode surface in order to get a picture of the behaviour of the water molecules. The simulations gave the scientists the opportunity to have a look at how water affected battery performance at the atomic scale and provided insights that were not possible with laboratory experiments alone. The simulations also found that a new electrolyte containing a lithium salt and an ionic liquid could separate and bind up water and sequester individual water molecules. As revealed by the computer simulations, preventing the water molecules from gathering into “puddles” and thus losing reactivity was an important parameter in the reaction. Through analysing the clustering of water molecules in the electrolyte matrix, the computer simulations were able to make out a quantitative relationship between the water tolerance of the electrolyte and the saltiness of its components. The water molecule could be trapped by the different ions at a specific range of concentration ratios in the salty medium which made it less reactive.
Solving problems related to water content in batteries has been among the chief interests of scientists around the world for quite some time. In 2020, a new electrode couple, LiV3O8-LiMn2O4, for aqueous Li-ion batteries was developed to understand the mechanism by which the water-in-salt electrolyte enhanced the cycling stability at an extended voltage window. Through operando synchrotron transmission x-ray microscopy on the LiMn2O4 cathode, the scientists showed that the water-in-salt electrolyte was able to avert the mechanical damage to the electrode network and dissolution of the electrode particles as well as decelerate the water decomposition process. As the viscosity of water-in-salt was higher, the reaction heterogeneity of the electrodes was analysed using x-ray absorption spectroscopic imaging. The study was aimed at finding a strategy to mitigate the possible kinetic limitations in three-dimensional battery architectures.
Image: Experimental setup for operando TXM imaging of LiV3O8-LiMn2O4 aqueous Li-ion batteries. (A) Schematic showing the experimental layout. CCD, charge-coupled device. (B) Operando cell design and key components. (C) Photo of the operando TXM experiment. Photo credit: Yu-chen Karen Chen-Wiegart, Stony Brook University and Brookhaven National Laboratory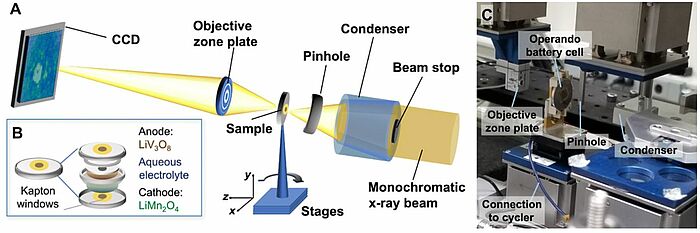
Source: Cheng-Hung Lin, Ke Sun, Mingyuan Ge, Lisa M. Housel, Alison H. McCarthy, Mallory N. Vila, Chonghang Zhao, Xianghui Xiao, Wah-Keat Lee, Kenneth J. Takeuchi, Esther S. Takeuchi, Amy C. Marschilok, and Yu-chen Karen Chen-Wiegart/ Systems-level investigation of aqueous batteries for understanding the benefit of water-in-salt electrolyte by synchrotron nanoimaging/ Science Advances Vol 6, Issue 10, 6 Mar 2020/ DOI: 10.1126/sciadv.aay7129/ Open Access This article is licensed under a Attribution-NonCommercial 4.0 International (CC BY-NC 4.0)
In 2021, scientists designed a rechargeable chloride-ion battery consisting of a water-in-salt electrolyte, a zinc anode, and a carbon cathode (graphene, carbon nanotubes, carbon black). These cathodes showed initial reversible specific capacities of 136, 108, and 102 mAh g⁻¹, respectively. Especially when graphene was used as the cathode, a reversible discharge capacity of 95 mAh g⁻¹ was retained after 2000 cycles. Also, the use of “water-in-salt” electrolytes was witnessed to enhance the discharge platform of aqueous chloride-ion batteries to 2.6V. The charge and discharge mechanism of the carbon cathode was analysed using transmission electron microscopes, Fourier-transform infrared spectroscopy, Raman spectroscopy, and X-ray photoelectron spectroscopy, which proved the reversible absorption/desorption of chloride ions in carbon cathodes.
Image: The reaction mechanism of CIB (A) Schematic representation of the rechargeable chloride ion battery. Herein, graphene, carbon black, and carbon nanotubes serve as cathode; Zn foil serves as cathode; and the saturated solutions of tetramethylammonium chloride was used as electrolyte. (B) The reaction mechanism of the negative electrode during the charging and discharging process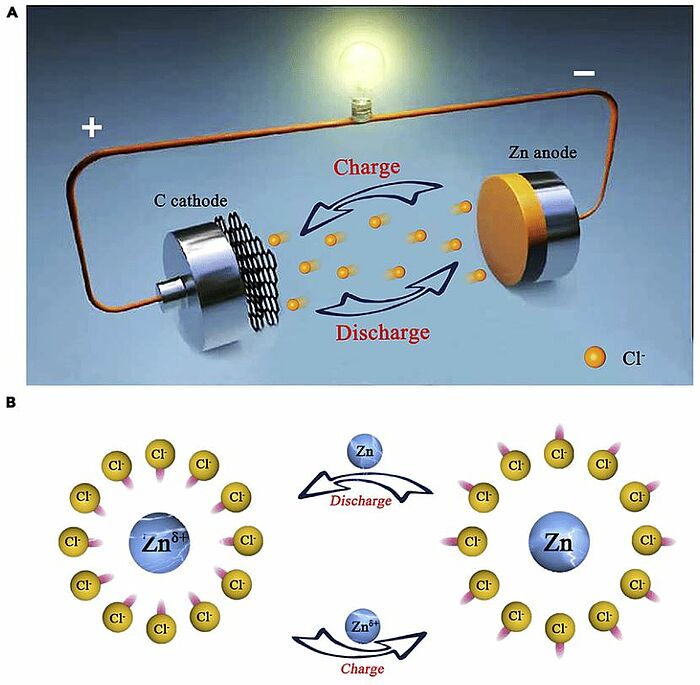
Source: Tong Li, Mingqiang Li, Hang Li, Hu Zhao/ High-voltage and long-lasting aqueous chlorine-ion battery by virtue of “water-in-salt” electrolyte/ iScience 24(1):101976, January 2021/ DOI:10.1016/j.isci.2020.101976/ Open Access This article is licensed under a Attribution 4.0 International (CC BY 4.0)
There are several advantages an electrolyte containing water holds: water-based electrolytes generate very high ionic conductivity, which is particularly suitable for high power density batteries. The production costs are lower and they are non-flammable. The novel electrolyte created by Argonne can contain much more water than the electrolytes currently used in electric vehicle and consumer batteries. This could help reduce costs in battery fabrication.
The study potentially provides whole new opportunities for battery manufacturers to include water in the battery fabrication process which enables cost reduction as well as more environmentally friendly manufacturing.
