Today, many research organisations are investigating new methods to achieve a clean transition from fossil energy to renewable fuels. The ETH Zurich and the German Aerospace Center (Deutsches Zentrum für Luft-und Raumfahrt; DLR) have discovered a thermochemical pathway to produce liquid fuel by means of concentrated solar energy. The SUN-to-LIQUID project, which is funded by the EU and Switzerland, has succeeded in synthesising solar kerosene from water and CO2 with concentrated sunlight.
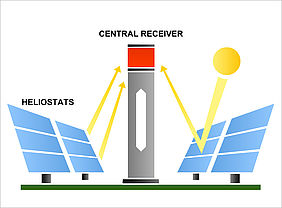
For this purpose, researchers at the German Aerospace Center scaled up existing technology for solar fuel production and carried out a unique experiment (2019) with a solar concentrating plant that was built at the IMDEA Energy Institute in Móstoles, Spain. There, a sun-tracking field of heliostats concentrated the sunlight by a factor of 2,500°C, which was three times greater than temperatures achieved at current solar power plants used for electricity generation. Through this concentrated solar energy reaction temperatures of more than 1,500°C could be reached within the solar reactor located at the top of the tower.
The solar reactor, developed by project partner ETH Zurich, generated synthesis gas from water and carbon dioxide by means of a thermochemical redox cycle. An on-site gas-to-liquid plant, which was developed by the project partner HyGear, then converted this gas to kerosene.
The German Aerospace Center can look back on many years of experience in researching solar-thermal chemical processes and their components. In 2014, the EU-funded SOLAR-JET project successfully demonstrated how renewable kerosene could be obtained directly from sunlight, water and carbon dioxide (CO2). The innovative process also used concentrated sunlight to convert carbon dioxide and water to a synthesis gas (syngas). This was achieved by means of a redox cycle with metal-oxide based materials at high temperatures. The syngas, a mixture of hydrogen and carbon monoxide, was then turned into kerosene by using Fischer-Tropsch technology.
In 2017, scientists at the German Aerospace Center generated syngas by concentrating the flux radiation of 3,000 ‘suns’ into a high-temperature thermal solar reactor at 1,500°C and causing thermochemical splitting of H2O and CO2 into H2 and CO. The experiment was carried out at ETH Zurich, Switzerland, as part of the five-partner SOLAR-JET project. In total, the experiment yielded 700 L of syngas after 295 consecutive cycles of the 4 kW solar reactor. This was then refined into kerosene by Shell Global Solutions in the Netherlands using the Fischer-Tropsch method. The most important component in the splitting process was a reticulated porous ceramic (RPC) structure in the reactor, which was made of a material called ceria (CeO2) that facilitated molecule splitting. This was the first experimental coupling of solar syngas production from H2O and CO2 with storage, compression, and FT-processing to liquid hydrocarbons.
The solar-driven kerosene production process has many advantages: compared to conventional fossil-derived jet fuel, the net carbon dioxide emissions to the atmosphere can be reduced by more than 90% with the new technology. Also, as the solar energy-driven process relies on water, CO2 and solar energy and does not compete with food production, it can meet the future fuel demand at a global scale without the need to replace the existing worldwide infrastructure for fuel distribution, storage, and utilisation. FT-processed kerosene, derived from H2O and CO2, can be certified for commercial aviation by a minor addendum to the existing D7566 specification for synthesized hydrocarbons. Furthermore, it can provide a secure and sustainable supply of renewable aviation fuel and more generally for transport applications.
SUN-to-LIQUID is a four-year project and is supported by the European Union’s Horizon 2020 research and innovation programme and the Swiss State Secretariat for Education, Research and Innovation (SERI). It began in January 2016 and will end on 31st December 2019.
Energy News Monitoring
New Sun-to-Liquid Process for Clean Kerosene Production