Aenert. Research Laboratory news
In search of an alternative technology for battery design sodium-ion batteries have emerged as potential candidates as a replacement for traditional lithium-ion batteries. Lithium-ion comprise lithium, cobalt, copper and nickel, materials which are scarce, have high environmental impact and high cost. Sodium on the other hand has a high natural abundance and is fairly low-cost.
Sodium-ion batteries are very similar to lithium-ion batteries but use sodium ions (Na+) as the charge carriers. Lately, there has been renewed interest in this technology as in view of the energy crisis the need for alternative or complementary battery technologies has become more urgent. Among the many anode materials which can be used in this type of battery anatase titanium dioxide (TiO2) is one of the most promising as it is low cost, non-toxic and of great abundance. However, its low electronic conductivity and slow ion diffusion kinetics at high rates have so far stood in the way of more widespread practical applications.
Now (2022), scientists the College of Materials Science and Engineering at Qingdao University in China have demonstrated a sol-gel approach, a method for producing solid materials from small molecules, to synthesise thermally stable anatase nanoparticles with a carbon shell as anode materials for sodium-ion batteries.
When the anatase that had been synthesized was tested in a half battery, the battery exhibited a reversible specific capacity of 228 mAh g-1 – at a current density of 0.05 Ag g-1 with 100% capacity retention after 2,000 cycles at 1 Ag-1, which was an astounding result.
Moreover, in-situ X-ray diffraction and Raman spectroscopy results showed a nearly zero-strain characteristic of anatase during charge/discharge processes. In-situ transmission electron microscopy, ex-situ X-ray photoelectron spectroscopy and scanning electron microscopy were also performed and the results showed that an irreversible sodiation-activation process occurred which formed a sodiated-TiO2 phase during the initial discharge process. A full coin cell assembled with anatase as the anode and Na3V2(PO4)3 as the cathode delivered an energy density of 220 Wh kg.
Image: (a) XRD patterns of H-XTiO2@C (X represents calcination temperatures). (b) Raman spectra of H-750TiO2@C and 750TiO2@C. (c) TGA curve and (d) nitrogen adsorption/desorption isotherms and DFT pore size distribution curve (the inset in (d)) of H-750TiO2@C. FESEM images of 750TiO2@C (e) and H-750TiO2@C (f). (g), (h) TEM images along with SAED pattern (the inset in (h)) of H-750TiO2@C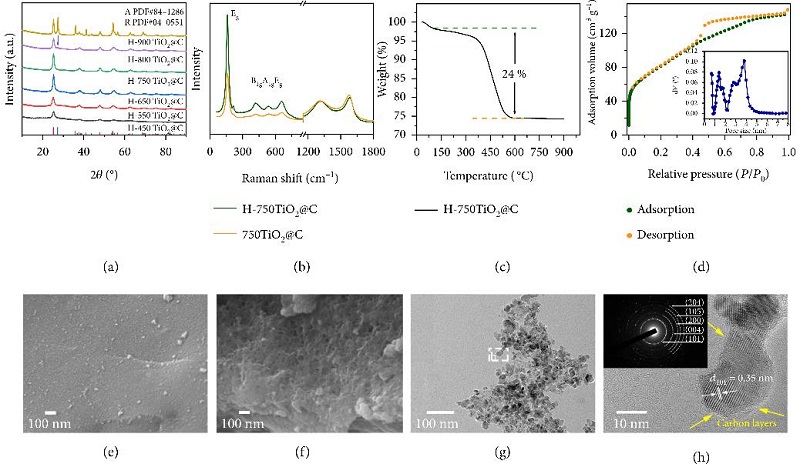
Source: Fujie Li, Chao Wang, and Xiu Song Zhao/ Sodium-Ion Storage Properties of Thermally Stable Anatase/ Energy Material Advances, Volume 2022, 06 Oct 2022/ doi.org/10.34133/2022/9876319/ Open Source This is an Open Access article is distributed under the terms of the Creative Commons Attribution 4.0 International (CC BY 4.0)
Finding new and cheaper methods for battery production has been at the centre of scientific research for several years. In 2017, scientists designed a sodium super-ionic conductor structured electrode, sodium vanadium titanium phosphate, which had a high specific capacity of 147 mA h g⁻¹ at a rate of 0.1 C and excellent capacity retentions at high rates. A symmetric sodium-ion full cell showed a superior rate capability with a specific capacity of about 49 mA h g⁻¹ at 20 C rate and ultralong lifetime over 10,000 cycles. Moreover, in situ synchrotron diffraction and X-ray absorption spectroscopy measurement were performed to get a better understanding of the underlying sodium storage mechanism and charge compensation behaviour. The results hinted at the potential application of symmetric batteries for electrochemical energy storage given the superior rate capability and long cycle life.
Image: Crystal structure of Na2VTi(PO4)3. (a) Schematic crystal structure of Na2VTi(PO4)3, and a-c represent different axes. (b) Rietveld refinement based on the synchrotron diffraction data. Black circle, red line and blue line represent the observed, calculated and difference patterns, respectively. The olive tick marks correspond to the Bragg reflections; the inset shows the TEM (scale bar, 100 nm) and HRTEM images (scale bar, 4 nm)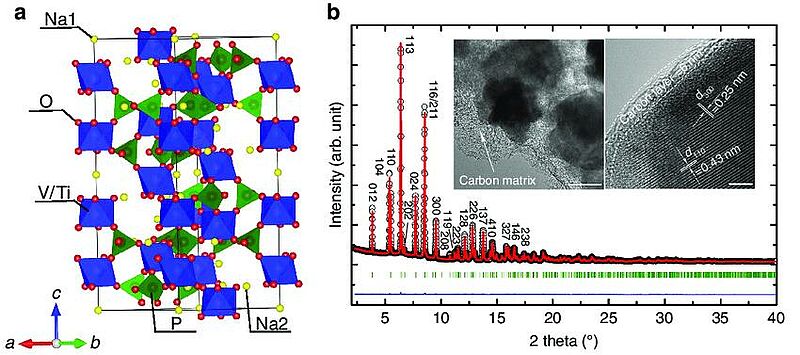
Source: Dongxue Wang, Xiaofei Bie, Qiang Fu, Ditty Dixon/ Sodium vanadium titanium phosphate electrode for symmetric sodium-ion batteries with high power and long lifespan/ Nature Communications 8(1):15888, June 2017/ DOI:10.1038/ncomms15888/ Open Source This is an Open Access article is distributed under the terms of the Creative Commons Attribution 4.0 International (CC BY 4.0)
In 2022, scientists proved that the multisodium storage in the polyanionic cathode could be improved and stabilised by increasing the entropy of the polyanionic host structure. The obtained polyanionic Na3.4Fe0.4Mn0.4V0.4Cr0.4Ti0.4(PO4)3 cathode showed multicationic redox property and could achieve high capacity with good reversibility under the high voltage of 4.5V (vs Na/Na⁺). Then they explored the underlying mechanism through operando characterizations and discovered a stable trigonal phase with reduced volume change during the multisodium storage process. Besides, the enhanced performance of the HE material also derives from the synergistic effect of the diverse TM species with suitable molarity. These results were proof of the effectiveness of high‐entropy concept in expediting high‐performance polyanionic cathodes discovery.
Image: Crystal phase of HE‐NASICON. a) Schematic illustration of the transition‐metals (TMs) distribution in NASICON structure. The gray and red balls represent P and O atoms, respectively. The balls with other colors represent different types of TMs. b) Rietveld refinement profile of the HE‐NASICON material. c) Schematic illustration of the crystal structure of HE‐NASICON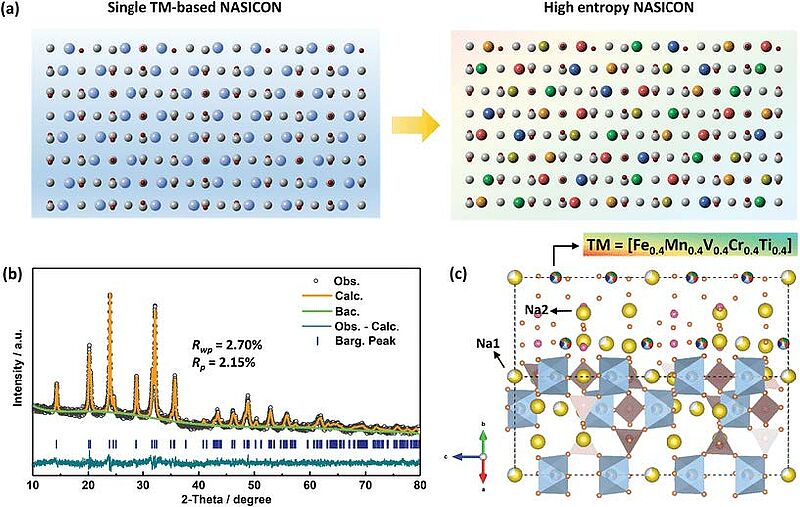
Source: Huangxu Li, Ming Xu, Huiwu Long, Jingqiang Zheng/ Stabilization of Multicationic Redox Chemistry in Polyanionic Cathode by Increasing Entropy/ Advanced Science 9(25), July 2022/ DOI:10.1002/advs.202202082/ Open Source This is an Open Access article is distributed under the terms of the Creative Commons Attribution 4.0 International (CC BY 4.0)
There are many benefits to be derived from using sodium-ion batteries: Although compared to lithium-ion batteries, current sodium-ion batteries have a slightly higher cost, slightly lower energy density, they exhibit better safety characteristics, and similar power delivery characteristics. If the cost of sodium-ion batteries can be further reduced, they can be used for grid-storage and home storage, where battery weight is not of such great importance. If, in addition to cost improvements, the energy density is increased, the batteries could be used for electric vehicles and power tools, and also any other application where lithium-ion batteries currently employed.
The abundance of sodium is thousand times greater than lithium. Therefore, this element can be easily mined in many countries around the globe. If the overall cost of extraction and purification of sodium is considered which is far lower, Na-ion cells can be 20%-40% cheaper. However, one challenge remains which consists in implementing the technology in large-scale installations. There is hope that the current research will prove a significant milestone in this direction.
By the Editorial Board