Aenert. Research Laboratory news
The water gas shift reaction (WGSR) is an exothermic reaction taking place between carbon monoxide and steam to form carbon dioxide and hydrogen. In typical industrial applications, the WGSR is conducted as a two-stage process: the high temperature stage performed over an iron-based catalyst in the temperature range 320 – 450°C; the low temperature stage with copper-based catalysts in the temperature range 150 – 250°C b being employed. There is no generally accepted reaction mechanism for the WGSR. It depends on whether it is being studied for high temperature or low temperature as well as on the catalyst type.
One of the main problems with regard to WGSR is catalyst deactivation. The main causes of catalysts deactivation are thermal sintering, sulphur poisoning, chloride poisoning. In addition to Fe-based catalysts for the HT-WGSR or Cu-based catalysts for the LT-WGSR, other catalysts such as nickel, cobalt, molybdenum, platinum, gold, rhodium, and ruthenium are used for the WGSR to mitigate some of the aforementioned problems. Also, during the water gas shift reaction such as in many other industrial processes, too, a lot of CO2 is emitted into the atmosphere.
Now (2022), TDA Research Inc. (TDA) has launched a project to develop an integrated water-gas shift (WGS) pre-combustion CO2 capture technology which will better reduce CO2 emissions from integrated gasification combined cycle (IGCC) power plants and coal-to-liquid plants. The project team consists of a joint venture of several institutions: The Gas Technology Institute (GTI), the University of California, Irvine (UCI), Indigo Power Systems LLC (Indigo), the National Carbon Capture Center (NCCC), Praxair, and CB&I Lummus Technology (CB&I). In the course of the project a gasification reactor will be constructed which uses computational fluid dynamics and kinetic modelling to improve CO2 removal and hydrogen recovery. TDA will be in charge of the project and perform the system design and sorbent production. The company will collaborate with GTI in the design and construction of a slipstream test unit. UCI and Indigo will undertake the process modelling and design and carry out the system and economic analyses. Praxair will support the field tests at the Praxair Facility in Tonawanda, NY.
The aim of the project is to demonstrate techno-economic viability of an integrated WGS catalyst/CO2 removal system for IGCC power plants and CTL plants. To achieve this goal, a high temperature PSA adsorbent will be used for CO2 removal above the dew point of the synthesis gas. Also, a commercial low temperature catalyst will be used for water-gas-shift as well as an improved heat management system. Moreover, CFD optimised reactors capable of managing the WGS exotherm while maintaining energy efficiency will be constructed. A mesoporous carbon with surface functional groups will be employed that can remove CO2 via physical adsorption. Also, CO2-surface interaction is strong enough to allow operation at elevated temperatures. Because CO2 is not bonded through a covalent bond, the energy input for regeneration is low. The heat of CO2 adsorption is 4.9 kcal/mol for TDA sorbent. Much higher IGCC efficiency can be achieved due to high temperature CO2 capture.
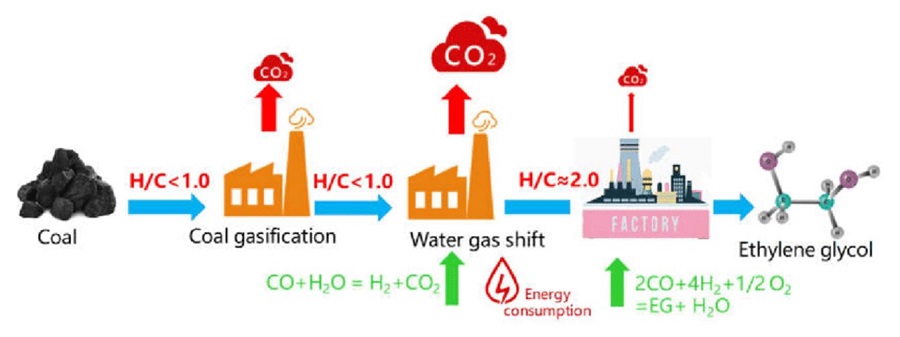
Utilising CO2 has long been at the centre of scientific attention for some time. In 2021, a study analysed the performance of four CO2 capture alternatives for the CTEG process: Rectisol, mono-ethanol amine (MEA), chilled ammonia process (CAP) and dimethyl carbonate (DMC) technologies. As shown by the results, the energy consumption of CO2 capture of the Rectisol process was the lowest, 1.88 GJ/tCO2, followed by the DMC process, the CAP process, and the MEA process. The CO2 capture cost of the Rectisol process was the lowest, CNY 169.5/tCO2, the DMC process – the second lowest, the CAP process was on third position, and the MEA process came last. As the Rectisol technology had the best comprehensive performance, it was the best option for CTEG industry in comparison with the MEA, CAP, and DMC technologies.
Image: Schematic diagram of the high CO2 emission reasons for coal to ethylene glycol (CTEG) process
Source: Yanqing Ma, Yitao Liao, Yi Su, Baojie Wang/ Comparative Investigation of Different CO2 Capture Technologies for Coal to Ethylene Glycol Process/ Processes 9(2):207, January 2021/ DOI:10.3390/pr9020207/ Open Source This is an Open Access article is distributed under the terms of the Creative Commons Attribution 4.0 International (CC BY 4.0)
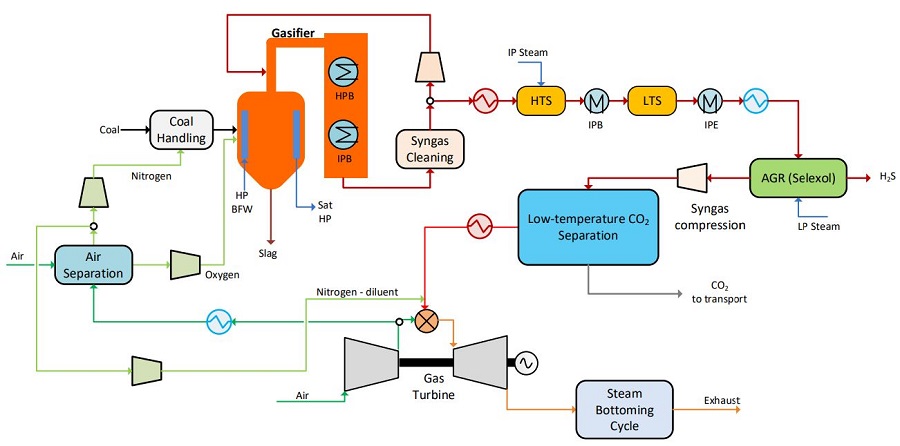
In 2022, scientists designed a process for low-temperature CO2 capture from an Integrated Gasification Combined Cycle (IGCC) power plant. Steady-state simulations were carried out and the performance of the overall process, as well as major process components, were looked at. For the baseline capture unit layout which produced high-pressure CO2 at 150 bar, the net specific power requirement was 273 kJe/kgCO2 and an 85% CO2 capture ratio was obtained. 12 different process parameters and their impacts were studied in a sensitivity analysis. The results showed that the highest impact on power requirements was made by compressor and expander efficiencies, as well as synthesis gas separation temperature. Modifying the process to producing cold liquid CO2 for ship transport resulted in 16% increase in net power requirements and is well suited for capturing CO2 for ship transport.
Improving the carbon footprint of IGCC and CTL technologies has several advantages: The new carbon capture technology is suitable for IGCC and coal-to-liquid plants that use a wide range of coals (bituminous to low-rank) and petcoke. TDA integrated a WGS pre-combustion CO2 control system in the installations which achieves a high CO2 capacity and removal efficiency (greater than 90 percent) at temperatures well above the syngas dew point for all commercial gasifiers. This high-temperature CO2 removal capability makes it superfluous to condense the steam in the gas and increases the mass flow through the gas turbine and also advances the efficiency of the power cycle. This technology will provide an effective carbon capture system that is much more efficient than conventional low temperature scrubbers.
Image: Block diagram of the integrated gasification combined cycle with low-temperature CO2 capture
Source: David Berstad, Geir Skaugen, Simon Roussanaly, Rahul Anantharaman/ CO2 Capture from IGCC by Low-Temperature Synthesis Gas Separation/ Energies 15(2):515, January 2022/ DOI:10.3390/en15020515/ Open Source This is an Open Access article is distributed under the terms of the Creative Commons Attribution 4.0 International (CC BY 4.0)
Carbon capture is believed to have the potential to play a key role in meeting climate change targets, providing low-carbon heat and power, decarbonising the industry and, as a more recent development, facilitating the net removal of CO2 from the atmosphere. The task that stands before us now is to advance existing technologies from the field-testing stage to large-scale implementation.
By the Editorial Board